Introduction
Reproduction is one of the most important aspects of a species’ biology, because it ensures the maintenance of population viability and the sustainability of the species in space and time (de Mérona et al., 2009). The reproductive cycle of a species involves a set of physiological and behavioural processes in relation to various environmental factors (Paugy et al., 2006). Indeed, reproductive parameters such as sex ratio, size at first maturity, fecundity and spawning period provide information which exhibit variations in response to environmental conditions (Neves et al., 2020; Santana et al., 2019; Souza et al., 2014). Thus, when variations in environmental conditions occur, changes in reproductive parameters also occur to ensure the success of the reproductive process. These variations within the initial reproductive pattern in response to environmental fluctuations are known as reproductive strategy (Bénech & Quensière, 1985).
In the natural environment, fish reproductive strategies have been extensively documented (Bailly et al., 2008; Espírito-Santo et al., 2013; Konan et al., 2014; Paugy, 2002; Wagaw et al., 2022). Several authors (Bailly et al., 2008; Paugy, 2002) agree on the synchronization between the reproduction of most fish species and the seasonal fluctuations of certain environmental variables which act on physiology by stimulating or inhibiting certain stages of gametogenesis or spawning. In temperate regions, the variables discriminated are temperature and photoperiod. Indeed, a strong relationship exists between reproductive cycles and variations in temperature and photoperiod (Abdulfatah, 2010; Migaud et al., 2002). In tropical environments, rain and floods are the main environmental factors inducing gametogenesis and egg-laying (Bénech & Quensière, 1985; Janáč et al., 2010). Indeed, the reproduction period of the majority of fish species coincides with the rainy season and the flood season in the tropical regions. The synchrony between major events in fish reproduction cycles and fluctuations in water levels has been the subject of much discussion (Gomes & Agostinho, 1997; Humphries et al., 1999). For many authors (Bailly et al., 2008; Bénech & Ouattara, 1990; Paugy, 2002), the coincidence of fish reproduction periods with the rainy and flood season can be explained by the establishment of favourable conditions in aquatic ecosystems during the rainy season. These favourable conditions result in water dilution, food abundance and access to refuge areas. In general, as floods in rivers are mainly due to precipitation, the evolution of these two climatic events overlaps. It is therefore not easy to separate the effects of rainfall and floods on the fish reproduction process. However, other authors believe that the first rains after the dry season are considered the stimulus for the formation of schools of long-distance migratory fish species and their subsequent movements (Agostinho et al., 2004), while floods play an essential role in stimulating spawning by promoting gonad maturation (Bailly et al., 2008). Based on the hypothesis that rainfall and floods have different effects on the biology of the fish, the present study attempted to understand the effect of rainfall on the one hand and the effect of water level on the other hand on the biology of Ctenopoma petherici. More specifically, this work attempts to answer the question: do precipitation and water level different effects on the reproduction of C. petherici? It also describes the spawning strategy of this species.
Material and Methods
In order to determine the role of each factor (rains and rising) in the C. petherici reproduction, two different ecosystems have been chosen: Ayame lake 2 and Agneby river (Fig. 1). These two reservoirs are located in the South-East of Côte d’Ivoire.
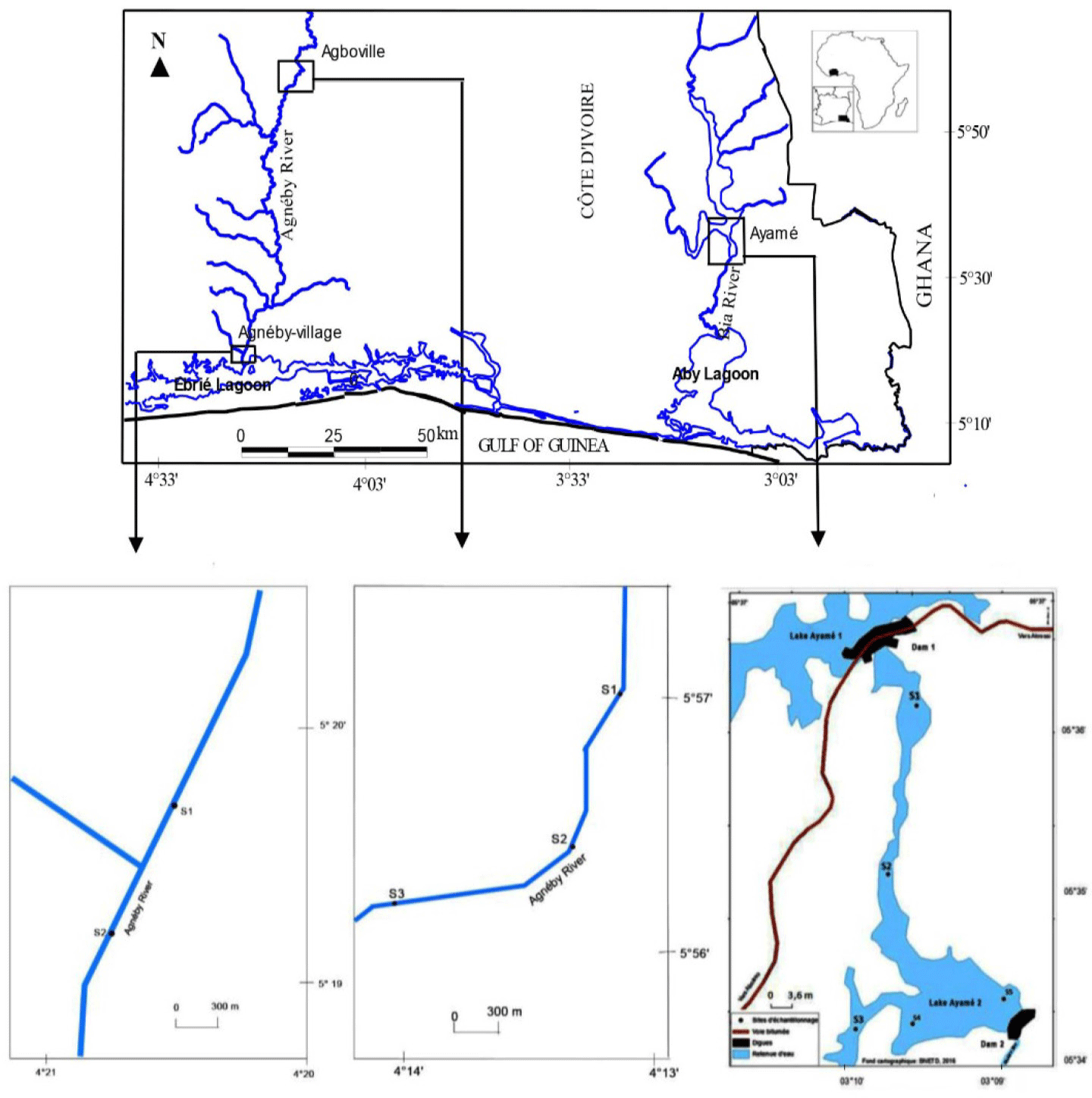
Ayame lake 2 and Agneby river are influenced by the equatorial transition climate, characterized by two rainy seasons separated by a short dry period from August to September, and a more pronounced dry season from December to March (Savané & Konaré, 2010).
The Ayame lake 2 hydroelectric dam (5°34´–5°37´N and 3°09´–3°10´W) was formed by the construction of two hydroelectric dams, Ayame 1 in 1959 and Ayame 2 in 1964 on the Bia River (da Costa et al., 2000). This aquatic ecosystem is located between dike 1 and dike 2 where water is permanent during the dry and rainy seasons. Thus, in the long dry season, the water level is maintained thanks to the closure of the dams’ spillways.
The Agneby river is a seasonal floodplain River characterized by strong seasonal fluctuations in water level. Indeed, during the long dry season, the water level drops considerably and during the rainy season; the flood is also significant.
Sampling occurred monthly over a 12-month period from September 2015 to August 2016. Fish came from experimental fisheries, then samples were completed by professional fishermen who practise the artisanal fishery at the sampling sites. The types of fishing gear used were gillnets (with a mesh size of 8 to 30 mm), traps and hooks. The collected specimens were identified according to Paugy et al. (2003). The fish were dissected and the gonads were examined macroscopically to determine the maturity stages of the ovaries (Table 1; Gogbé et al., 2017). The gonads and liver were removed and weighted to the nearest 0.001 g, then the body weight was determined to the nearest 1 g.
Data from Gogbé et al. (2017).
Percentage of mature females: The percentage of mature females (PMF) is expressed by following relation: PMF = Nmf / Nf × 100 where Nmf is mature female number and Nf the captured female number.
Gonado-somatic index: The gonado-somatic index (GSI) was estimated by:
GSI = GW / EW × 100 where GW = gonad weight (g) and EW = eviscerated fish weight (g).
Hepato-somatic index: The hepato-somatic index (HSI) was evaluated using:
HSI = LW / EW × 100 where LW = liver weight (g) and EW (g).
Effect of rain and flood on gonad maturation: In this section, the influence of the water level on the state of ripening was first of all checked. The influence of rain on the state of maturation was minimised or even eliminated by taking only observations from the long dry season (December to March). Thus, the lake represents the high water level and the river the low water level. Then, in order to show whether precipitation and high water levels have similar or different effects on maturity, we considered the data from the lake’s long dry season for the high water level, thus subtracting the influence of precipitation, and the transition period (March to April) between the long dry season and the long rainy season in the river. This choice is justified by the fact that the first rains fall during this period, but the water level in the river remains low.
Spawning strategy: Histological investigation of ovaries and determination of proportions of oocytes in different stages of development is an appropriate method for the description of spawning characteristics (Lefler et al., 2008). Indeed, to determine the spawning strategy of C. petherici, the homogeneity of the ovaries at different stages of development was evaluated. This evaluation consisted of examining microscopic sections taken at each stage of ovarian development and determining the proportion of different categories of oocytes.
Two variations of the Chi-square test have been used. Chi-square test of independence (χ2) was used for the “relationship between water level and fish maturity” and “influence of rainfall and high water level on fish maturity. For the relationship between water level and fish maturity, the null hypothesis H0 was defined as follows: the state of maturity and water level are independent. The alternative hypothesis H1 is: fish maturity depends on water level. For the effect of rainfall and high water on the maturity of fish, the null hypothesis H0 is as follows: rainfall and high water have the same effect on the maturity of fish. Alternative hypothesis: rainfall and high water levels different effects on fish maturity.
for χ2obs > χ2ex: H0 is rejected;
for χ2obs < χ2ex: expected, H0 is accepted;
with χ2obs = χ2 observed and χ2ex = χ2 expected.
To compare the proportions of mature females between the wet and dry seasons, the Chi-square test for comparing two proportions was used.
The Mann-whitney test was used to compare the GSI between the rainy season and the dry season. The tests were carried out at a confidence level of 95%.
Results
Breeding season was determined on the basis of occurrence of most advanced sexual maturity stages (stage III, IV and V; Fig. 2) and of the GSI (Fig. 3) of females.
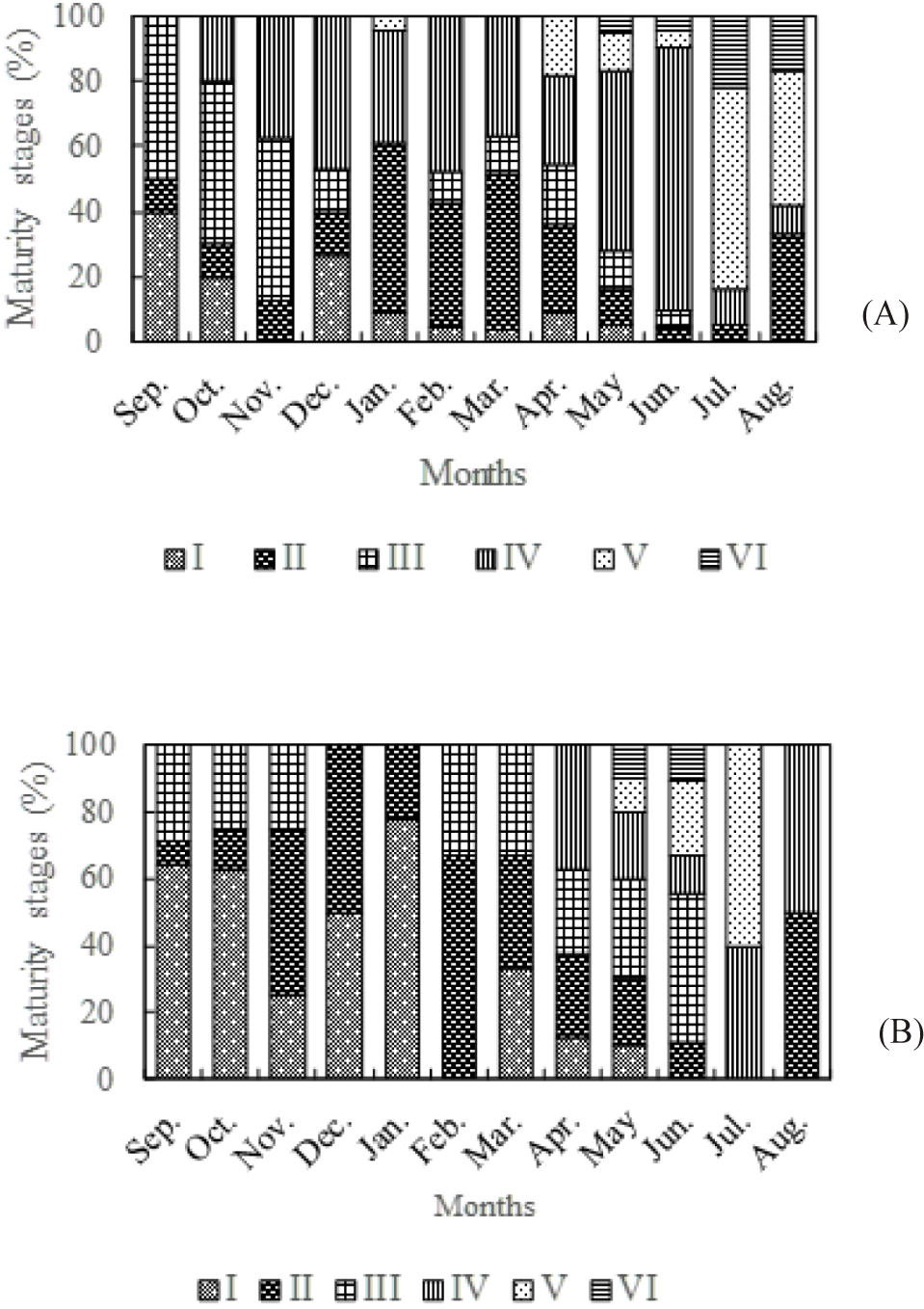
In the Ayame lake 2 (Fig. 2A), most advanced sexual maturity stages were observed throughout the year. However, the proportion of ripe gonads is much higher during the long rainy season from May to June. During this period, mature gonads represent more than 80% of the gonads observed. The proportion of ripe gonads drops during the long dry season, particularly during the month of January, with less than 40% of gonads observed.
In the Agneby river (Fig. 2B), females at advanced sexual maturity stages were examined over two periods: September to November and February to June. Pre-spawn females (stage IV) appear in the catches from April to August while females at the stage V were caught during the period from May to July with a maximum of 60% in July. Although females at stage III of sexual maturation were recorded during the periods from September to November and from February to March, the proportion of immature females (stage I and II) caught during these same periods is high (superior at 50%). The notable fact is that no ripe gonads were observed during the low flow from December to January.
For both environments, the proportion of mature gonads was significantly higher in the rainy season (Chi-square test, p < 0.05).
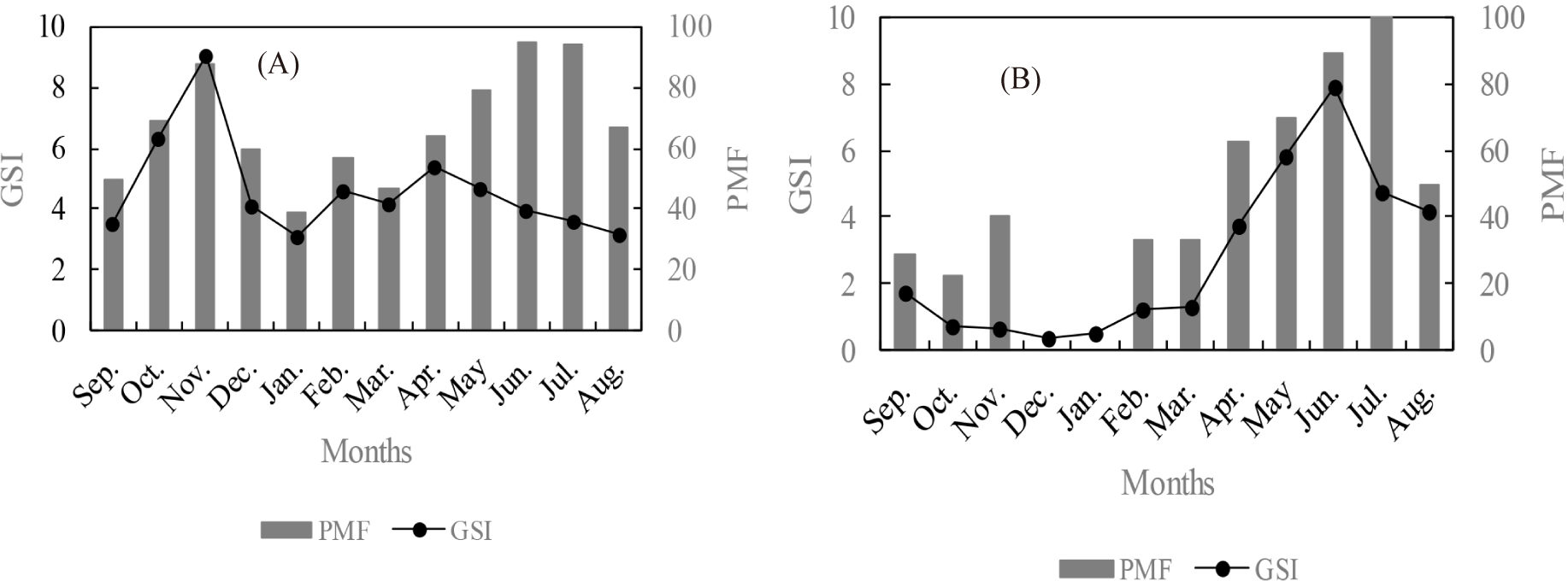
Monitoring of the mean monthly GSI of C. petherici females shows that in the Ayame lake 2, this parameter ranges from 3.13 in January to 9.06 in November. It has two peaks: the first (9.06) was recorded in November and the second (5.43) was recorded in April. Thus, the monthly mean GSI indicates two continuous cycles of gonad development in females of C. petherici in Ayame lake 2. A short cycle that is between September and December and a long cycle from January to August (Fig. 3A). During these different cycles of gonad development, the PMF is high (> 50%). The monthly mean GSI indicates continuous reproduction of C. petherici in dry season and rainy season in the Ayame lake 2.
In contrary to the phenomenon observed in the Ayame lake 2, the variation of the monthly mean GSI has two major phases in the Agnéby River (Fig. 3B). The first phase, during which low GSI values were noted, extends from September to March. During this period, the GSI oscillates between 0.39 (December) and 1.76 (September). The lowest proportions of mature females (< 50%) were also noted during this same period with a total absence of mature females in December and January. The second phase begins in April and ends in August. During this period, the monthly average GSI values are higher and fluctuate between 3.76 in April and 7.89 in June. It corresponds to the gonad development cycle characterized by a growth phase of the GSI from March to June and a decay phase from June to September.
GSI is high during the rainy season than during the dry season, both in the lake and in the river (Mann-Whitney U test; p < 0.05).
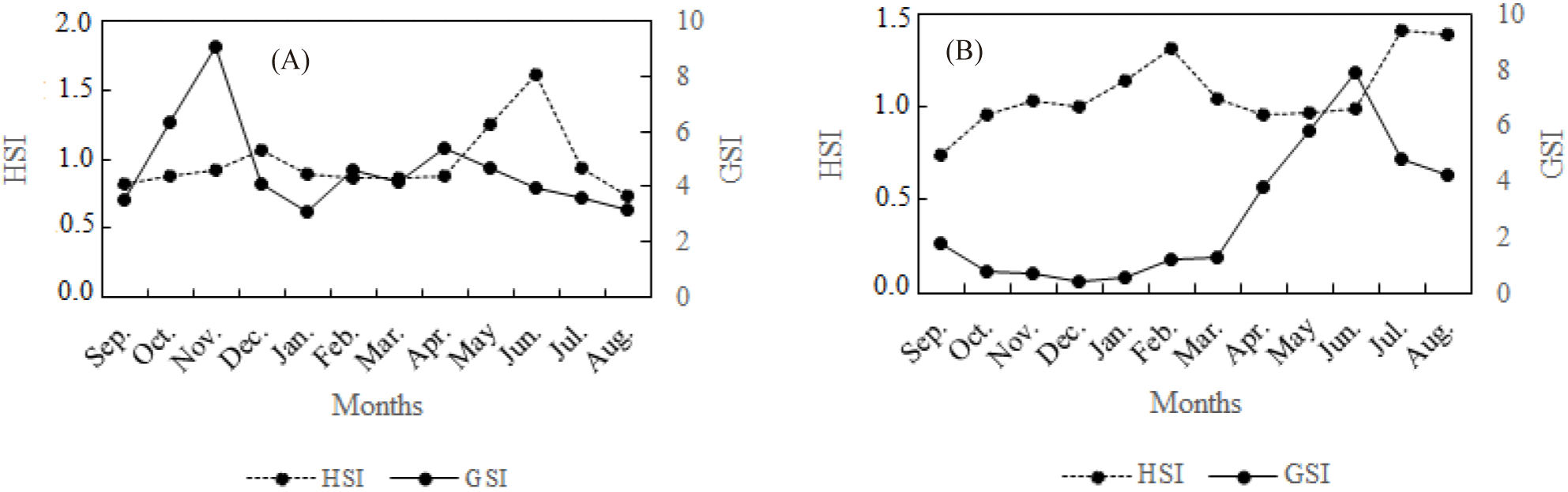
Analysis of the monthly evolution HSI shows that the peaks 1.07 and 1.62 were recorded in December and in June respectively in Ayame lake 2 (Fig. 4). These months correspond to the different periods during which the GSI decreases in the females examined. Thus, as the GSI increases, the HSI gradually decreases. This is most noticeable in the Agneby river, where while the GSI is at a low level (September to February), the HSI grows and reaches its first peak (1.31%) in February just before the start of growth in the GSI. When GSI begins to grow from March, the HSI decreases proportionally before starting a new increase in June.
The analysis of the state of maturity on the basis of the water level shows that 50% and 9.52% of the females are mature in the high water and in the low water respectively. These values subjected to the Chi-square test of independence (χ2obs = 11.34, p < 0.05) show that the water level has a significant influence on the reproduction of C. petherici. Concerning the effect of rain and water level on the state of maturity, the Chi-square test (χ2obs = 0.08, p > 0.05) did not reveal any significant difference between the proportions of mature females depending on these two parameters. Indeed, for the period defined to maximize the effect of rain and minimize that of the water level, a proportion of 54.54% of mature females was observed compared to 50% in high water. This result allows us to reject the null hypothesis according to which rainfall and water level would have different effects on C. petherici maturity.
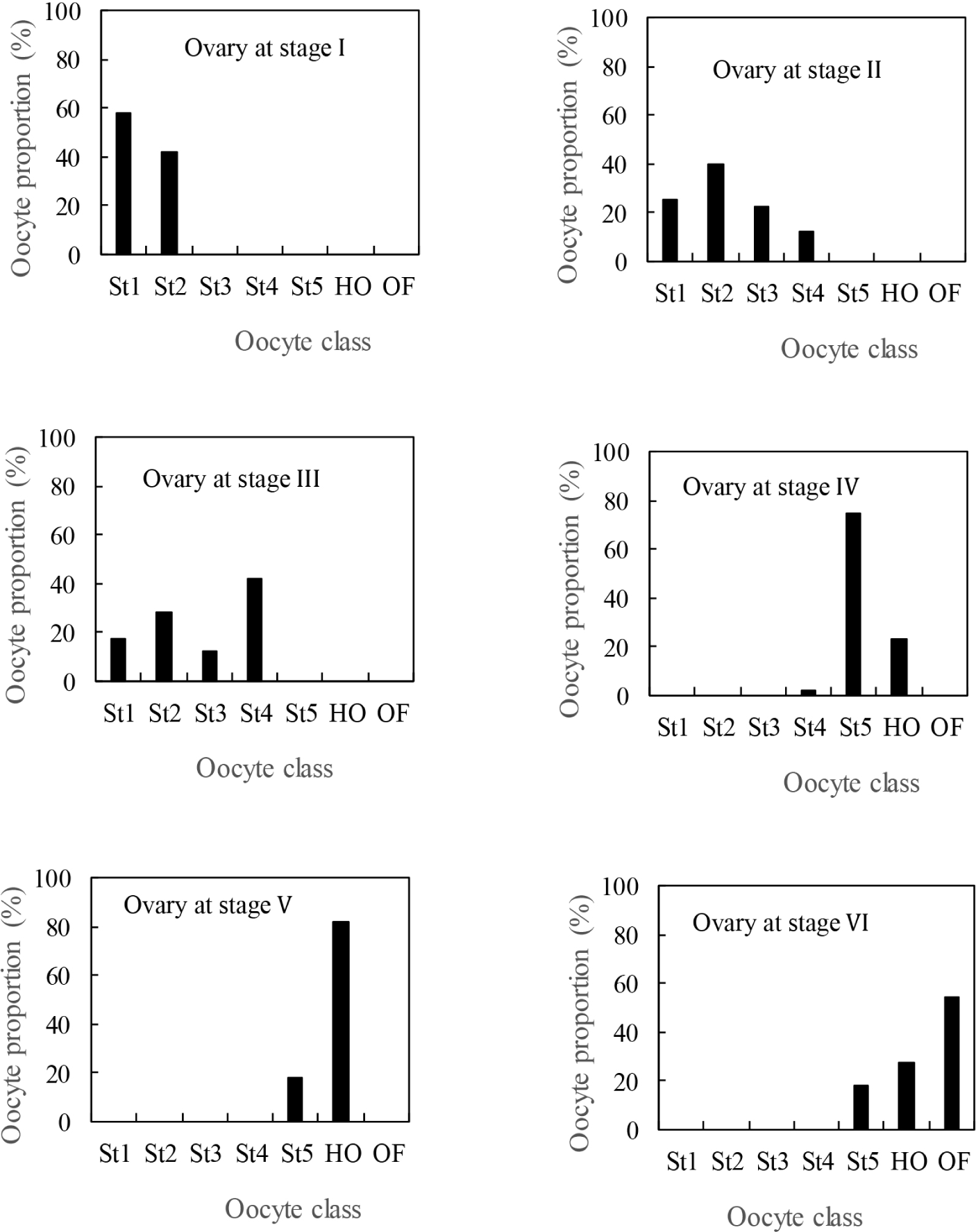
Fig. 5 shows the proportion of different categories of oocytes at each stage of ovarian development. In stage I of development, the structure of the ovary consists of immature oocytes divided into two classes of oocytes: plurinucleolar oocytes and perinucleolar oocytes. At stages II and III of maturity, the structure of the ovary is heterogeneous with different classes of oocytes (previtellogenic and vitellogenic oocytes). At the pre-laying and laying stages, only mature oocytes (ripe oocytes and hydrated oocytes) are observed in the ovary. However, oocyte classes, each consisting of a stage of oocyte maturity in the ovary, show a generally unimodal oocyte population at each stage of the ovarian cycle. This expresses a homogeneity of the ovary in C. petherici, particularly at the time of spawning.
Discussion
Monthly analysis of stages of sexual maturation, PMF and GSI give indications on the gonad maturation cycle and the breeding season. In the Agneby river, data indicate that gonad maturation in C. petherici is seasonal. Thus, the breeding season of C. petherici in the Agneby river covers the period from April to July which corresponds to the long rainy season. Similar results were obtained by Boussou et al. (2005) on Synodontis shall in the same river. The synchronization of the breeding season and the rainy season in the rivers is a well-known strategy for many tropical fish species (Adite et al., 2005; Paugy, 2002). The influence of rain on fish reproduction could be due to its effect on the physico-chemistry of water (dilution of dissolved salts; Bénech & Ouattara, 1990) and on the abundance of food resources (Konan et al., 2013, 2014). This strategy allows juveniles not only to have shelters in submerged areas with the arrival of the rains but also to have available food resources (Konan et al., 2013, 2014; Oso et al., 2011). In first analysis, the data obtained in the river show that the rains and the flood would be the main factors inducing gametogenesis in C. petherici.
Furthermore, monitoring of GSI, stages of sexual maturity and PMF allows us to deduce that C. petherici reproductive activity occurred practically throughout the year in Ayame lake 2. Reproductive investment was also higher during the rainy season. In order to understand the maintenance of reproductive activity during the dry season in the lake, on the one hand, and its more intense level during the rainy season, on the other hand, we tried to dissociate the effect of rain from that of the amount of water present in the environment. The results of these analyses indicate that the arrival of rain stimulates gonad maturation in the same way as the presence of a significant and permanent amount of water in the environment. Thus, despite the lack of rain during the dry season, the maintenance of reproductive activity in the lake is attributable to the significant amount of water in this ecosystem. Many authors have reported the importance of the water level in the reproductive activity of fish. For example, Santana et al. (2019) observed that water level fluctuations are of great importance for the population dynamics of two Characiformes species in the southern part of the Brazilian Pantanal. For Bailly et al. (2008), a high water level ensures the maintenance of favourable conditions for reproductive activity. These authors report that the high water level, particularly in lentic environments as is the case of Ayame lake 2, is a guarantee of stability. Indeed, the species tend to be less affected during reproduction due to the low variability of environmental conditions.
In view of the results obtained, the intensification of sexual activity during the rainy season can be explained by the combination of the effects of rain and the rise in water levels, two factors that stimulate the maturation of the gonads. The absence of sexual activity in the Agneby river during the dry season is due to the significant drop in water level. Indeed, the drop in water level can be considered as a stress factor and it is known that stress most often triggers a negative response to the reproductive success of males and females (Chehade et al., 2015; Schreck, 2010).
According to Bénech & Quensière (1985), the majority of fish reproduction strategies can be classified into two main types. The first is based on the spread of spawning over time and the second is based on the seasonal maturation of the gonads which generally takes place in a single annual spawning. However, the data collected in the Agneby river and Lake Ayamé 2 do not immediately allow us to classify C. petherici in one or the other of the two reproduction groups. This situation required additional data such as the spawning strategy to say whether the reproductive strategy of C. petherici follows a spread of spawning over time as observed in the lake or is based on the seasonal maturation of the gonads as in the river. For C. petherici, this analysis suggests that spawning is total and no fractionated. Indeed, the distribution of oocytes at the different stages of maturity indicates a unimodal pattern with the absence of immature oocytes at the most advanced stages of sexual maturity (stages IV and V). Thus, the type of spawning observed shows that the reproductive pattern of C. petherici is indeed based on the seasonal maturation of the gonads. Indeed, reproduction throughout the year in the lake attests that the permanence of water stimulates gametogenesis in C. petherici.
The maturation of fish gonads is accompanied by energy expenditure from either visceral organs such as the liver, muscles, or ingested foods (Delahunty & de Vlaming, 1980). The data presented by Koné (2000) and Atse et al. (2009) indicate that there is a relationship between the gonado-somatic and HSI when mobilized energy for gonad maturation is from the liver. The comparative analysis of the monthly evolution of these two parameters (GSI and HSI) in C. petherici revealed a negative correlation between these two parameters in this species in Lake Ayamé 2 and in the Agnéby River. This negative correlation indicates the use of liver reserves during gonad maturation (Bouhali et al., 2015). These results indicate that C. petherici is a lean fish.
Conclusion
The simultaneous analysis of the stages of sexual maturity, GSI and PMF in Lake Ayame 2 and in the Agneby river allows us to conclude that C. petherici reproduces all year round in the lake and mainly during the rainy season in the river. Precipitation and the large amount of water in the environment have the same effect on the maturation of the gonads of C. petherici. Our results indicate that rainfall and high water levels are two factors that stimulate gametogenesis and gonad maturation in C. petherici. The combined effect of these two factors induces the intensification of sexual activity. However, additional analyses are necessary to assess the influence of these two factors on fertility, oocyte quality and juvenile viability.
