Introduction
Vitamin E is a group of lipid-soluble molecules, including four tocopherols (α-, β-, γ- và δ-tocopherols) and four tocotrienols (α-, β-, γ- và δ- tocotrienols), in which α-tocopherol has the highest biological activity (Lee & Han, 2018). Vitamin E plays an important role in various physiological and biochemical processes in fish (Hamre, 2010; Lu et al., 2016). The antioxidant of vitamin E is essential to prevent oxidation by reactive oxygen species (ROS) in the lipids of cell membranes (Naderi et al., 2019). However, fish cannot synthesize vitamin E and they have to uptake it from the food to meet the physiological requirements. Optimum levels of vitamin E are recorded to improve growth (Liu et al., 2014; Zhang et al., 2021), enhanced immunity (Lu et al., 2016) and increased fish flesh quality (Zanon et al., 2018).
Fish depend on vitamin E dietary supply to fulfill their requirement (Bera et al., 2022). The optimum vitamin E requirements in diets are essential for normal fish growth and disease defense. Therefore, the optimal vitamin E levels have been determined for most farmed fish species including gilthead seabream (Sparus aurata; Montero et al., 2001), grouper (Epinephelus malabaricus; Lin & Shiau, 2005), red drum (Sciaenops ocellatus; Peng & Gatlin, 2009), cobia (Rachycentron canadum; Zhou et al., 2013), yellow croaker (Larmichthys crocea; Wang et al., 2016), pufferfish (Takifugu obscurus; Cheng et al., 2018), largemouth bass (Micropterus salmoides; Li et al., 2018) and snubnose pompano (Trachinotus ovatus; Zhang et al., 2021).
Snubnose pompano Trachinotus blochii is a commonly cultured marine fish in Southeast Asian countries, including Vietnam, owing to its fast growth rate, good meat quality and high market demand (Baliao et al., 2023; Thuy et al., 2024). T. blochii are cultured in open sea or brackish water cages and in ponds (Baliao et al., 2023). In industrial aquaculture, T. blochii must cope with stressors, particularly handling, and crowding. These factors can cause oxidant damage, influence the growth, immune response and ultimately the survival of farmed. Thus, dietary antioxidant supplementation such as vitamin E is essential.
In Viet Nam, aquaculture of this fish has expanded rapidly during the last decade. Few previous studies regarding their nutritional requirements have been reported (Chau & Ngo, 2015; Hung et al., 2013; Nguyen & van Do, 2021; Nguyen et al., 2021; Pham et al., 2021, 2022). These studies have shown the effects of different nutrient supplementation or replacement in diets on growth performance, feed utilization, and physiological response in this fish, studies regarded the effects of vitamin E supplementation on growth performance, whole body composition, and innate immune response in the fish juveniles are limited. Therefore, this study aimed to determine the effects of vitamin E supplementation on the growth, whole-body composition, and innate immune response in snubnose pompano juveniles.
Materials and Methods
In accordance with Vietnam’s National Regulations on the Use of Animals in Research (Decree 32/2006/ND-CP, 2006), snubnose pompano is not classified under either Group IB (endangered and critically endangered species) or Group IIB (threatened and rare species). As such, conducting this study did not necessitate obtaining a permit or ethical approval. Nonetheless, the researchers adhered to the highest standards of ethical practice in their treatment of animals throughout the study.
Snubnose pompano juveniles were obtained from a private hatchery (Nha Trang - Khanh Hoa Province) and transported to the Aquatic Disease and Seeds Research Central – Nha Trang University. Fish were acclimatized to laboratory conditions and fed the basal diet for 2 weeks. To start the feeding trials, fish (initial body weight of 5.22 ± 0.28 g) of similar size and no clinical signs of diseases were weighed. Twenty individuals were randomly assigned into one of 18 × 200 L composite tanks, and 3 replicates per treatment. The feeding trial lasted 10 weeks.
Experimental tanks were placed indoors. Sea water was supplied in a flowthrough system with a sand filter placed at the inlet. All fish groups were fed ad libitum four times daily at around 7:00, 11:00, 14:00, and 17:00. All tanks were continuously aerated and cleaned daily in the afternoon by siphoning off accumulated waste materials. About 50% of water in each aquarium was replaced daily. During the experimental period, temperature ranged from 25.5°C to 28.5°C, pH was 7.5–8.1, salinity fluctuated from 28–30 PSU, and dissolved oxygen was not lower than 6.5 mg/L.
We used the source of vitamin E from the alpha tocopherol acetate (50% alpha tocopherol acetate, Sigma-Aldrich, St. Louis, MO, USA). The feed ingredient composition used for basal diet was provided in Table 1. The basal diet (see details in Table 2) was supplemented with one of five vitamin E levels (100, 200, 400, 800, and 1,600 mg/kg diet). Basal diet (no vitamin E supplement) was used as the control feed.
3) Vitamin premix: Vitamin A, 1,000, 000 UI; Vitamin D3, 300.000 IU, Vitamin C monophosphate 10.000 mg; Vitamin B6, 500 mg; Vitamin B2, 320 mg; Vitamin B12, 5 mg (Provimi Vietnam, Bien Hoa City, Vietnam). Mineral premix: Zn (ZnO), 4.750 mg; Mn (MnSO4.H2O), 1.900 mg; Mg (MgO), 1.050 mg; Ca (CaHPO4.2H2O), 0.8%; Co (CoCO3), 47.5 mg; Se (Na2SeO2), 47.5 mg (Provimi Vietnam).
To prepare the feed, we mixed well all ingredients until the feed was homogenous in a Hobart-type mixer. Subsequently, we added lipids and water to the feed. Cold-extruded pellets (diameters of 1.0 mm and 2.0 mm) were produced and then air-dried. Pellets were vacuum-packed in plastic bags (approximately 100 g/kg moisture per bag), sealed and stored in the freezer at –20°C until being used to feed the fish juveniles. The vitamin E levels in the feed were 2.31 (control), 117.25, 189.03, 382.1, 758.54, and 1,437.32 mg/kg, respectively (analyzed by high-performance liquid chromatography).
At the end of the experiment, juveniles were fasted for 24 h. Subsequently, fish were anesthetized with monophenyl ether glycol (150–200 ppm). All juveniles in each tank were counted to estimate the survival (%) as the number of surviving individuals in each tank × 100% / 20. Fish were weighed and to calculate weight gain rate (WGR) as the mean of the final fish weight (g/fish) minus the mean of the original fish weight (g) in each tank. The relative increase in the weight gain or Percent WG (%) = (final mean weight − initial mean weight) / initial mean weight × 100. The specific growth rate (SGR, % per day) was calculated as ([Ln final mean weight − Ln initial mean weight] / test days) × 100. We also estimated the feed conversion ratio (FCR) by dividing the amount of feed intake (g)/wet weight gain (g).
Three individuals were randomly selected from each tank to measure the body weight, liver weight, and visceral mass weight for calculation of the hepatosomatic index (HSI) and viscerosomatic index (VSI). Similarly, another subset of three individuals per tank was collected, minced together and stored at −20°C for proximate composition analysis of the whole body.
We used the official method of the Association of Official Analytical Chemists (AOAC, 1990) to analyze the proximate composition in the diets and in the fish. To measure moisture content, samples were dried in an incubator (UNE-600, Memmert, Nuremberg, Germany) at 105°C until the weight was constant. To measure ash content, samples were combusted in an electric furnance (SH-FU-5MG, SH Scientific, Sejong, Korea) at 550°C for 6 h. Kjeldahl method was used to determine the crude protein content (AOAC, 1990) and the Soxhlet extraction method was used to measure the crude lipid content (AOAC, 1990).
Blood samples from three fish per tank were collected from the caudal vein. A portion of the blood was collected for hematological parameters analysis, and the rest was transferred to an Eppendorf tube overnight at 4°C before being centrifuged to collect the serum. Fish serum was stored at –80°C for later lysozyme activity analysis.
Haemoglobin (Hb), haematocrit (Hct), red blood cells (RBCs), and white blood cells (WBCs) were analyzed using the Sysmex XT-1800i Blood Analyzer (Sysmex, Hyogo, Japan).
Lysozyme activity in pompano serum was determined as described by Shugar (1952) with adjustment. Accordingly, 25 μL of the serum sample to be tested was placed into 1 well of a flat-bottomed 96-well plate. Add 175 μL of Micrococcus lysodeiltikus 0.075% bacterial suspension to the wells, mix rapidly and place the plate on a spectrophotometer (Titertek multiskan). Measure optical density at 450 nm every 30 s, continuously for 5 min. Lysozyme activity in pompano serum was determined according to the standard curve of the degradability of Micrococcus lysodeikticus (Sigma-Aldrich) of lysozyme extracted from egg white (white hen egg lysozyme, Sigma-Aldrich) in 6 wells as standard, containing 25 μL of lysozyme in decreasing concentrations of 20, 10, 5, 2.5, 1.25, and 0.625 μg/mL was prepared.
Protein efficiency ratio (PER) = weight gain/protein intake
Survival rate (SR, %) = (final number of fish – initial number of fish / initial number of fish) × 100
Viscerosomatic index (VSI, %) = (viscera weight / final body weight) × 100
Hepatosomatic index (HSI, %) = (liver weight / final body weight) × 100
Data are presented as means±SE and were analyzed using one-way ANOVA in SPSS (version 22.0, IBM, Armonk, NY, USA). Group comparisons were performed using Duncan’s multiple range tests. Significant differences were considered at p-values < 0.05. Prior to statistical analyses, raw data were checked for normal distribution and homogeneity of variance with the Levene’s test. Broken-line analysis based on specific growth rate and feed conversion ratio was used to determine the breakpoint that represents the optimum dietary vitamin E requirement of fish.
Results
The growth performance, survival, and morphological index of fish fed various experimental diets over 70 days are presented in Table 3. Remarkably, all snubnose pompano maintained a 100% survival rate throughout the study, with no death recorded. The addition of dietary vitamin E did not significantly influence the survival of snubnose pompano juveniles across different vitamin E levels over the 70-day duration (p > 0.05). Fish fed with diets containing 382.1 and 758.54 mg/kg of vitamin E also showed enhanced growth metrics, though to a lesser extent, while the lowest growth performance was recorded in fish on the vitamin E-free diet (p > 0.05). To use the broken line with SGR data indicated that the optimal level for adding vitamin E in food for pompano was 135.63 mg/kg (Fig. 1A).
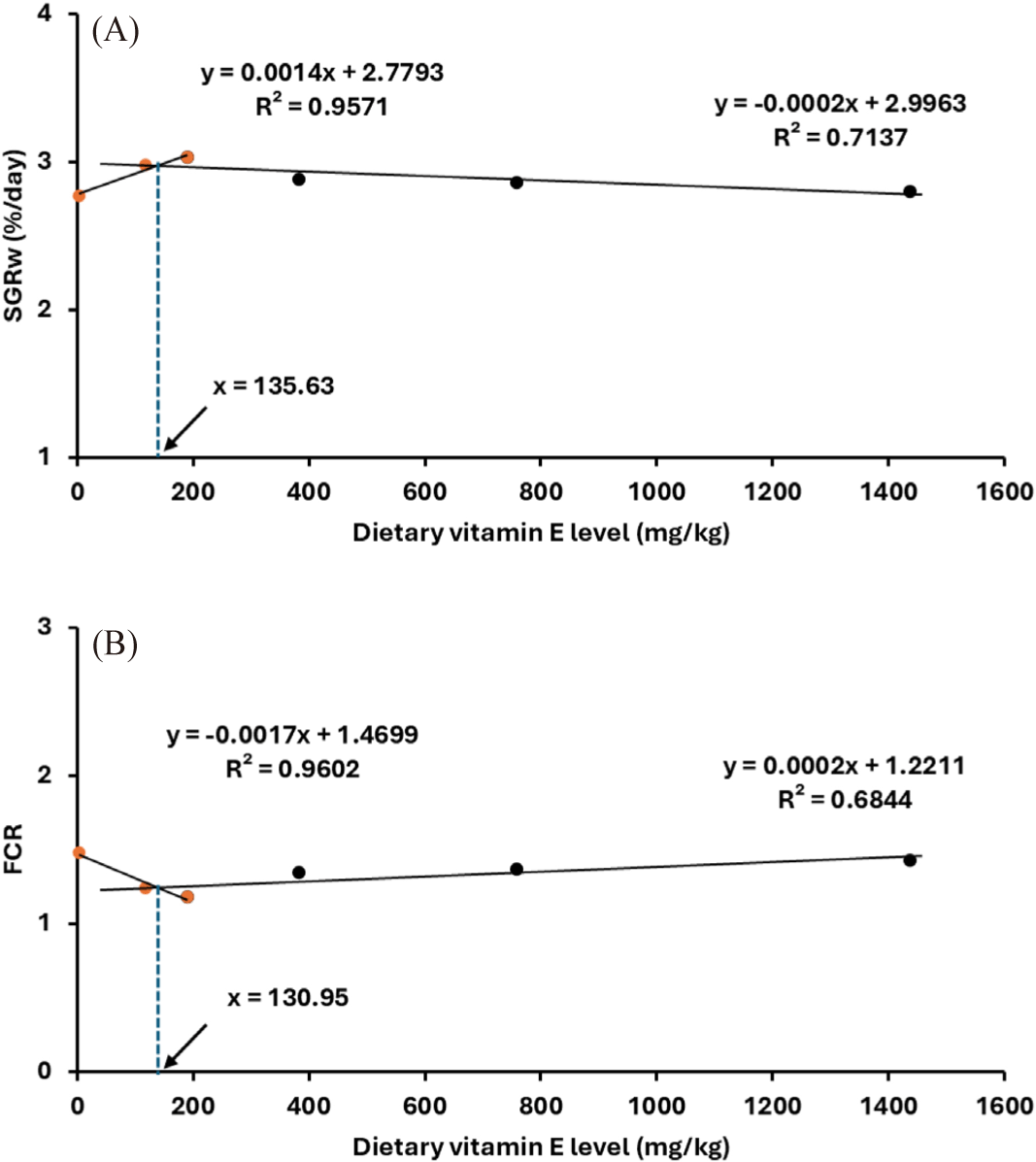
The FCR and PER were not significantly affected by the levels of dietary vitamin E. However, fish consuming 189.03 mg/kg of vitamin E exhibited a lower FCR and higher PER in comparison to those on the basal diet. Interestingly, the broken line analysis using FCR data indicated that the optimal level for adding vitamin E in food for pompano was 130.95 mg/kg (Fig. 1B).
Furthermore, fish on the basal diet displayed significantly lower VSI and HSI indices than those receiving vitamin E-supplemented diets. Nevertheless, there were no significant differences in VSI and HSI among the groups fed with vitamin E-enhanced diets.
The addition of vitamin E had a significant impact (p < 0.05) on the whole-body proximate composition of the fish, as detailed in Table 4. The lipid, protein, and ash content in fish fed with vitamin E-enriched diets were significantly higher than those observed in fish on the control diet. Notably, the highest values for these parameters were found in fish receiving diets containing 117.25 and 189.03 mg/kg of vitamin E. This positive trend was also reflected in the moisture content of the fish.
Following the ten-week experiment period, significant differences were observed in the WBC count, Hb, Hct, and lysozyme activity among snubnose pompano fed diets supplemented with vitamin E compared to those on the basal diet (p < 0.05, Fig. 2). The highest WBC counts and lysozyme levels were recorded in the fish receiving 382.1 mg/kg of vitamin E, while the lowest values were found in those on the basal diet.
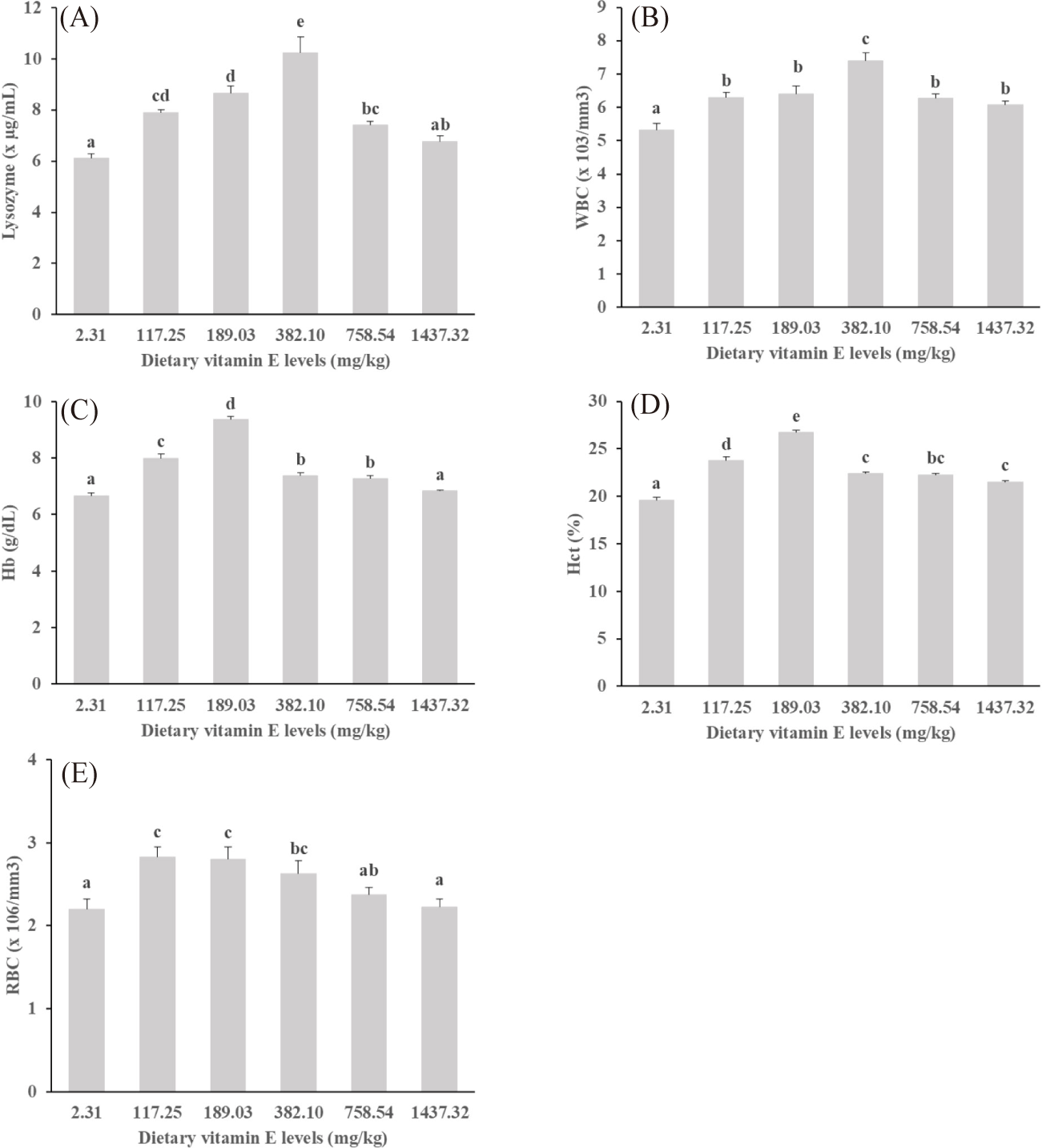
The count of RBC was not affected by dietary vitamin E levels (p > 0.05). However, significant impacts were noted on Hb and Hct levels (p < 0.05), with the highest values of Hb and Hct observed in fish fed a diet supplemented with 189.03 mg/kg of vitamin E. In contrast, the lowest values were noted in fish on the basal diet.
Discussion
Our study on the effects of dietary vitamin E on snubnose pompano demonstrates marked improvements in growth performance, body composition, and immune response, while also achieving a consistent 100% survival rate throughout the experimental period. These results highlight the beneficial impact of vitamin E supplementation, a trend that has been observed across various marine fish species, albeit with species-specific variations in optimal dietary levels. Incorporating vitamin E into the diets of aquatic animals helps prevent the oxidative rancidity of unsaturated fatty acids in their diets and tissues, supporting normal metabolism. This, in turn, enhances feed efficiency, protein uptake, and overall growth performance (Qiang et al., 2019). Specifically, our findings indicated that fish fed diets with vitamin E supplementation showed significantly greater final weights, weight gains, and specific growth rates compared to those on a diet without vitamin E supplementation. Furthermore, a reduction in the FCR and an improvement in the PER were noted in fish receiving 189.03 mg/kg of vitamin E, suggesting that the optimized FCR and enhanced PER are key factors in promoting growth efficiency.
In our study, the determined optimal level of vitamin E for the growth of snubnose pompano was found to exceed those identified in other species such as red drum (S. ocellatus) at 31 mg/kg (Peng & Gatlin, 2009), cobia (R. canadum) at 78 mg/kg (Zhou et al., 2013), largemouth bass (M. salmoides) at 73 mg/kg (Li et al., 2018) and another study on snubnose pompano (T. ovatus) at 90.75 mg/kg (Zhang et al., 2021). However, it was lower than the levels recommended for Japanese eel (Anguilla japonica) at 212.9 mg/kg (Shahkar et al., 2018), gilthead seabream (S. aurata) at 2,900 mg/kg (Atalah et al., 2011), and meagre (Argyrosomus regius) at 451 mg/kg (Lozano et al., 2017). Conversely, several studies have reported that growth performance in fish was not significantly influenced by dietary vitamin E concentrations (Montero et al., 2001; Moreau & Dabrowski, 2003; Tocher et al., 2002), suggesting variability in response to vitamin E. These differences could stem from factors specific to each species, such as the size of the fish, the composition and palatability of the diet, feeding frequency and quantity, as well as the conditions under which experiments were conducted.
Vitamin is widely recognized as a crucial nutrient for promoting growth and is commonly recommended as a daily supplement in fish diets. However, it is important to note that an overdose of vitamin E in the feed can be toxic to fish and potentially hinder their growth, as highlighted in some studies (Paul et al., 2004). In our investigation, we observed a decline in the growth performance of snubnose pompano when subjected to diets with excessively high levels of vitamin E supplementation. This suggests that beyond a certain threshold, additional vitamin E may be preferentially utilized to bolster the immune system rather than to support further growth. This finding underscores the necessity of balancing vitamin E supplementation to optimize both health and growth outcomes in aquaculture practices.
The variation in dietary vitamin E levels did not influence the survival rates of fish over a 10-week experimental period, a finding consistent with observations in other species such as white sturgeon (Acipenser transmontanus; Moreau & Dabrowski, 2003), sub-yearling beluga (Huso huso L.; Amlashi et al., 2011), and golden pompano (T. ovatus; Zhang et al., 2021). This alignment across different studies, including our own, indicates that snubnose pompano, alongside the species mentioned, is capable of withstanding short-term vitamin E deficiency without adverse effects on survival. This resilience suggests that while vitamin E is essential for optimal health and growth, these fish species possess a degree of tolerance to its deficiency in the short term.
The ratios of specific organ or tissue masses to total body mass, such as the condition factor, VSI, and HSI, serve as indicators of changes in nutritional and energy status (Goede, 1990). Additionally, the liver is recognized as the primary storage site for α-tocopherol, a component of vitamin E (Hamre, 2010). In our study, fish that were fed diets supplemented with vitamin E exhibited significantly higher HSI and VSI compared to those receiving the basal diet without vitamin E supplementation. This observation aligns with findings from studies on sub-yearling beluga (H. huso L.; Amlashi et al., 2011) and yellow catfish (Pelteobagrus fulvidraco; Lu et al., 2016), suggesting a common response among different fish species to dietary vitamin E. The observed increase in HSI in conjunction with higher dietary levels of vitamin E may reflect an enhanced deposition of vitamin E in the liver and an increase in energy reserves, indicating a beneficial effect of vitamin E supplementation on organ health and nutritional status (Amlashi et al., 2011). While most vitamins function primarily as cofactors, vitamin E is recognized for its role as a powerful antioxidant. A deficiency in vitamin E can lead to oxidative cell damage, resulting in severe fat malabsorption and muscle atrophy (Watanabe et al., 1997). In the present experiment, supplementing the diet with vitamin E significantly affected the protein, lipid, ash, and water content in the whole body. These findings are consistent with previous studies (Saheli et al., 2021; Sau et al., 2004).
Blood parameters, including hemoglobin (Hb), hematocrit (Hct), red blood cells (RBC), and white blood cells (leucocytes), are indicative of health and immune response in fish (Zhou et al., 2013). In our investigation, the addition of vitamin E did not alter RBC counts, yet significant differences were observed in Hct and Hb levels. Notably, a vitamin E-deficient basal diet resulted in a marked reduction in Hb and Hct percentages in snubnose pompano, suggesting that insufficient vitamin E may either inhibit hemoglobin synthesis or accelerate its breakdown (Hamre & Lie, 1995). Our findings align with research conducted on channel catfish, (Ictalurus punctatus; Yildirim-Aksoy et al., 2008), cobia (Zhou et al., 2013), and hybrid striped bass (Li et al., 2018), demonstrating similar impacts of vitamin E on hematological parameters. Conversely, several studies have reported higher Hct and Hb values in fish not supplemented with vitamin E (Naderi et al., 2019; Qiang et al., 2019; Yildirim-Aksoy et al., 2008), indicating potential species-specific responses or differences in experimental conditions. Additionally, research on sub-yearling beluga (H. huso L.; Amlashi et al., 2011) and gilthead seabream (S. aurata; Montero et al., 2001) found no significant effects of dietary vitamin E on hematological parameters, further suggesting variability based on factors such as experiment duration, fish species, and size. The discrepancies observed across studies may be attributed to these varying conditions, emphasizing the need for tailored dietary recommendations to optimize the health and immune response of different fish species. This underscores the complexity of nutritional requirements in aquaculture and the importance of species-specific research to inform dietary formulations that support optimal fish health and performance.
Leukocytes and lysozyme play pivotal roles in the non-specific immune system of fish, functioning as key components in the defense against pathogens (Amlashi et al., 2011). Lysozyme, a mucolytic enzyme of leukocytic origin, target the peptidoglycan layers of Gram-positive bacteria by cleaving the β (1–4) linkages between N-acetylmuramic acid and N-acetylglucosamine, resulting in bacterial lysis (Saurabh & Sahoo, 2008). Past research has established that dietary vitamin E is capable of bolstering the immune response in various fish species, showcasing its importance in aquaculture nutrition (Lu et al., 2016; Puangkaew et al., 2004; Zhou et al., 2013). Our findings contribute to this body of evidence, illustrating that a deficiency in vitamin E within the basal diet markedly reduces both WBC count and lysozyme activity. Conversely, supplementation with higher doses of vitamin E (382.1 mg/kg) was associated with increased WBC counts and lysozyme activity, underscoring vitamin E’s efficacy in enhancing immune responses in snubnose pompano. This effect highlights the crucial role of vitamin E not only in supporting growth and health but also in fortifying the innate immune system, further emphasizing the need for appropriate dietary formulations that incorporate adequate levels of vitamin E to ensure the well-being and disease resistance of cultured fish.
In summary, our research demonstrates that suitable supplementation with vitamin E significantly improves growth performance, whole-body composition, and natural immune responses in juvenile snubnose pompano. An optimal vitamin E dosage of 117.25–189.03 mg/kg in the diet is identified for enhancing growth and body composition, hemoglobin, and hematocrit, whereas a higher dose of 382.1 mg/kg is recommended to boost the natural immune response through increasing lysozyme activity and WBC. These findings underscore the critical role of vitamin E in aquaculture nutrition, supporting the health and development of snubnose pompano.
For future research, it would be beneficial to explore the long-term effects of vitamin E supplementation on snubnose pompano, particularly focusing on the potential impacts on reproductive performance and lifespan. Additionally, investigating the interactions between vitamin E and other dietary components (such as vitamin C, selenium, fatty acids) could offer insights into more holistic dietary strategies. The role of vitamin E in stress resistance and disease prevention under commercial farming conditions also warrants further examination. Such studies could provide a deeper understanding of the mechanisms behind vitamin E’s benefits and inform more refined dietary recommendations that optimize the health, growth, and overall productivity of snubnose pompano in aquaculture operations.







