Introduction
Shrimp aquaculture has grown to be a significant sector in East and Southeast Asian developing nations, accounting for the majority of exports in terms of both volume and value. Currently, Asian nations produce more than 70% of the shrimp in the world, and the majority of that output is exported to support the economies of those nations (Venkateswarlu et al., 2019). One of the world’s top producers of shrimps is Bangladesh.
In order to maintain the ideal conditions for shrimp growth, water quality management mainly entails managing certain water quality parameters on a daily basis. The regulation of water quality is a key factor in shrimp farming since poor water quality hinders shrimp development and survival. To prevent environmental stress on shrimp that might hasten the development of many diseases, water quality management is crucial (Tharavathy, 2014). And aquatic plant plays a vital role to maintain water quality of a water body.
Macrophytes are crucial for the organization of aquatic environments (Meerhoff et al., 2003). They act as the principal source of primary production in pond and lake ecosystems, feeding aquatic species and providing them with breeding grounds and habitats for foraging. This helps to sustain the diversity and stability of aquatic organism assemblages (Agostinho et al., 2007).
Most research concentrate on the ecological functions of submerged plants, and growing and floating leaved species, which are numerous in eutrophic turbid water but relation to shrimp growth have received less attention (Cazzanelli et al., 2008; Rozas & Odum, 1988).
It is common in central and southern Europe, northern and tropical Africa, and Asia. Najas indica is an aquatic, submerged, invasive plant that thrives in fresh water (calm waters such as rivers, slow-flowing aquatic environments, ponds, and lakes). Najas gramina and Ruppia maritima typically grow in large numbers in Asian countries with salinities between 10 and 0 ppt (Krishna, 1988). It is typically found between 0.6 and 4.5 m deep, loves somewhat warm water (8 degrees Celsius at its coldest), but can also survive in the hot summer weather. A 40 cm long, branching stem is seen and the leaves are opposite, slender and toothed, up to 2 cm long It truly grows from seeds each spring, making it an annual. Between July and September, seeds develop in the axils of leaves (Filzgerald, 1969). N. indica grows abundantly in the southwest part of Bangladesh. However, the use of N. indica in aquaculture has not yet been tested. The purpose of this study is to look into how N. indica effects on the survival, growth rates of tiger shrimp and as well as water quality.
Material and Methods
The experiment was conducted at nine ponds located within the pond complex of the shrimp research station in Bagerhat, Bangladesh Fisheries Research Institute (BFRI). This Institute situated in 10 km east from Sixty Dome Mosque (a UNESCO world heritage site). The exact coordinates of these ponds (22˚38˝26.9′N–89˚48˝ 20.1′E).
In the experiment there was a control pond that did not have any N. indica, and two treatments, T1 and T2, were set up with 20% and 40% of the total water area covered by N. indica, respectively. Using the same climatic conditions and a three-month culture period (April–June, 2022). Stocking density of shrimp was 4 ind./m2. It compared the growth of shrimp (Penaeous monodon) at different percentage of N. indica containing ponds (gher). Used nine rectangular ponds with an average area of 0.1 hectare, and it duplicated each group three times in each.
In order to maximizing the ability of hydrogen sulfide to oxidize and to get rid of other unpleasant gases, the water from ponds was entirely drained out and re-excavated with dyke renovation before being allowed to sun dry for 7–10 days. Following liming (quick lime:dolomite 1:1) at a rate of 250 kg/ha of soil. Tidal water sourced from the neighboring Daratana River (22˚64˝45.9′N–89˚80˝15.7.1′E) was introduced into the pond, maintaining a depth of 1 m. This was achieved by using a Monk gate and exchanging the water twice throughout the entire cultivation period. Rotenone was used to get rid of undesired fish at a rate of 36.5 kg/ha. Urea and TSP were used to fertilize the water at 2.5 and 3.0 ppm, respectively, to hasten the change of the water’s color and the growth of plankton in the waterbody (Suresh et al., 2006).
Messrs emon fish of Rampal, Bagerhat district, Bangladesh, was the vendor from where the shrimp post larvae (PL) were purchased. The average weight and total length of each shrimp PL were roughly 0.01 ± 0.03 g (Mean ± SE) and 1.50 ± 0.02 cm (Mean ± SE) respectively, using a measuring scale and a digital electronic balance (Model WLC 1/A2, Radwag, Radom, Poland) and age of PL was 3 days. Afterwards, during the conditioning process, 10 L circular PL containers were filled with pond water and let to acclimatize to the salinity for an hour. Lastly, the PL was randomly distributed among all of the ponds in line with the experimental design.
Commercial pellet diets (C.P. Bangladesh, Gazipur, Bangladesh) were used in this study, specifically designed for P. monodon. These diets contain 12% moisture, 35% crude protein, 5% crude fat, 5% crude fiber, 18% ash, 26% carbohydrate, 2.5% calcium, and 1.4% phosphorus. This choice was made because marine shrimp have a higher protein requirement in their diet compared to freshwater prawns (Mantoan et al., 2021). Starter and grower feed were the most commonly used substances in ponds. The feeding schedule during the culture period was as follows: in week 1, nursery feed was applied at 100% body weight (BW) per day, in week 2, starter feed was applied at 60% BW, in week 3, starter feed was applied at 40% BW, in week 4, starter feed was applied at 10% BW, and finally, grower feed was applied at 2%–2.5% till the culture period was over (Ramaswamy et al., 2013). The complete quantity of feed was divided into two equal portions and given daily at 6:00 am and 6:00 pm. To measure the length and weight of the shrimp, 5% of each treatment were sampled every two weeks. Afterwards, according changes in body weight were used to adjust the meal levels. The feed conversion ratio (FCR) was calculated by keeping track of the food that was given.
To identify the species of the aquatic submerged plant of N. indica, the whole plant was sent to Bangladesh national herbarium, Mirpur, Dhaka 1216, Bangladesh. Accession number of N. indica was DACB 90649.
Water quality variables viz. temperature (°C), dissolved oxygen (DO) (mg/L) concentration, pH, total alkalinity (mg/L), salinity (ppt), conductivity (mS/cm), total dissolved solids and ammonia (mg/L) were measured at weekly intervals in the morning between 08:00 and 09:00 am. The temperature of water surface was recorded on-site using a conventional centigrade thermometer. A Secchi disc was used to record transparency, and a titrimetric method was applied to measure total alkalinity (APHA, 2008). HANNA Instruments, Romania’s HI3826 ammonia test kit was used to measure the ammonia levels. The rest of the parameters were measured using HQ40d multimeter (HACH, Loveland, CO, USA) made in US.
Following the conclusion of the cultivation period, the ponds were entirely drained in order to harvest all of the shrimp at once. Shrimp were subsequently measured, weighed, and counted for each replication. The parameters that were taken into account in order to ascertain the growth return, yield, and survival of the experimental shrimp were as follows: net weight gain (NWG), specific growth rate (SGR), absolute growth rate (AGR), FCR, gross fish yield, and survival rate as follows Equation (1)–(5) (Sutradhar et al., 2021).
SPSS software version 20.0 (Chicago, IL, USA) and Excel 2019 (Microsoft, Redmond, WA, USA) were used to analyze the data. The use of descriptive statistics, which provides a standard degree of precision like mean and standard deviation, was used to highlight the essential qualities. To find a significant difference, Duncan’s multiple range test was followed by an analysis of variance (ANOVA; p < 0.05). The relationship between shrimp growth and physico-chemical factors was investigated using correlation and multiple regression analysis.
Results
In Table 1, the mean values (Mean ± SE) for the experimental ponds’ water quality parameters during the culture period are listed. Fig. 1 shows the growth increase of shrimp from all ponds during the grow-out period at intervals of 15 days. The findings showed that all other parameters were statistically different between the control pond and ponds harboring N. indica, and that the water quality characteristics of culture ponds were significantly varied (p > 0.05) across triplicates in DO. But in case of temperature, there was no significant variations. Additionally, for all treatments, differences in growth performance between triplicates were negligible.
1) Data from Matias et al. (2002).

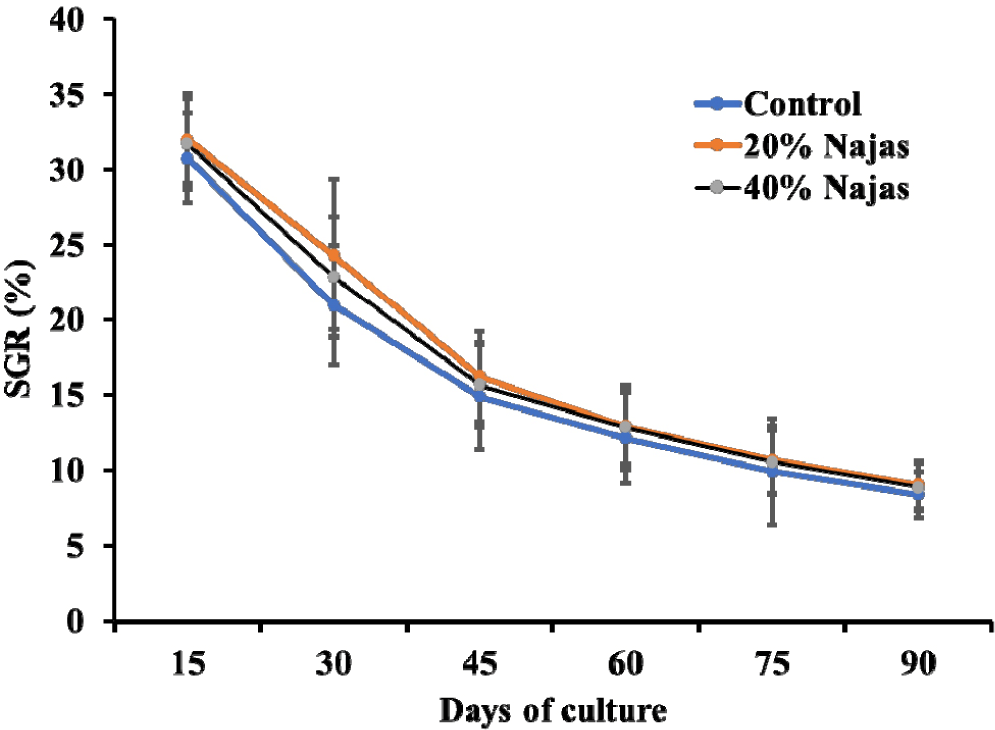
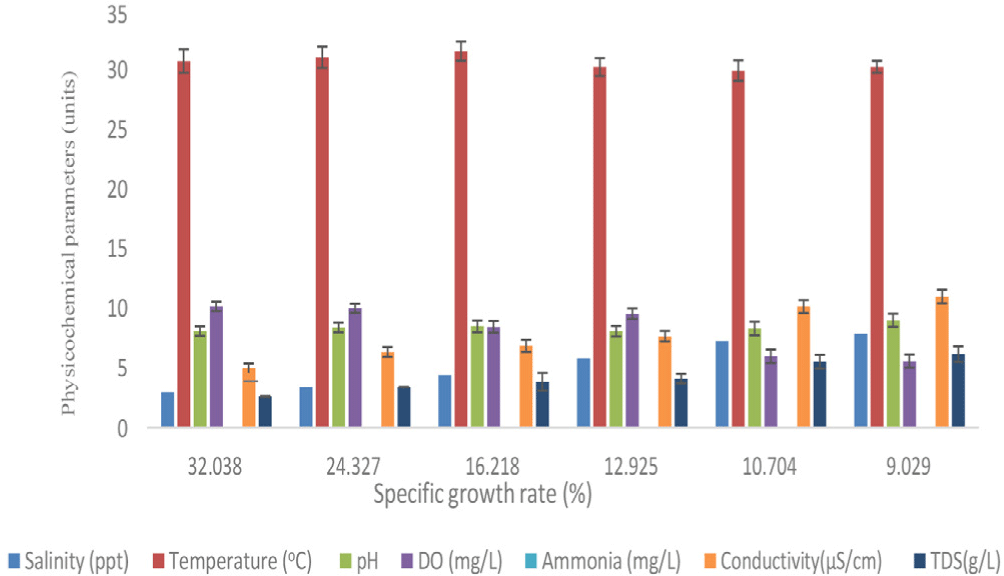
Table 2 provides the expected growth parameters for each treatment. The T1 pond (30.44 ± 0.77 g) and control (17.38 ± 0.63 g) were found to have the greatest and lowest final mean BWs, respectively. The SGR of all treatments were significantly different, although net weight increase displayed the same aptitude (F = 4,006.88, p = 0.000). SGR showed persistently reduced patterns as the rearing period lengthened (Fig. 2). However, there was a strong correlation between shrimp length and weight for all treatments (Table 3). With the ANOVA test, the total body length of all treatments was shown to be insignificant (F = 3,612.27, p = 0.000). The SGR ranged from 8.4 to 9.02% each day at the end of the culture period. In T1 pond, the highest apparent SGR of 9.02% per day was reached under the following water quality criteria: Salinity was 5.9 ± 0.73 ppt, the temperature was 30.91 ± 1.03°C, DO was 8.55 ± 1.06 mg/L, pH was 8.55 ± 0.47, and ammonia was 0.02 mg/L (Fig. 3). Significantly (F = 19.524, p = 0.002) higher survival rate (68.4%) was discovered in T1, although control had the highest mean FCR (1.51; p < 0.05). According to statistical analysis, T1 produced significantly larger gross production than that of the other ponds (832.8 ± 7.4 kg/ha/90 days) and that differed significantly (p < 0.05) among the three treatments (F = 4.001, p = 0.079) (Table 2).
| Treatment | Parameters of length-weight relationship | |||
|---|---|---|---|---|
| ‘a’ | ‘b’ | r | Remarks | |
| Control | 0.0026 | 3.37 | 0.98 | > 3: is growth exponent ‘b’ value that shows allometric growth |
| T1 | 0.003 | 3.26 | 0.96 | |
| T2 | 0.0027 | 3.33 | 0.91 | |
While mean shrimp weight showed a negative correlation with DO (r = –0.822, p = 0.002), alkalinity (r = –0.876, p = 0.000), and temperature (r = –0.744, p = 0.009), it had a strong positive correlation with mean length (r =0.853, p = 0.001), salinity (r = 0.947, p = 0.000), TDS (r = 0.924 p = 0.000), conductivity (r = 0.956, p = 0.000), ammonia (r = 0.551, p = 0.079) and pH (r = 0.121, p = 0.724) (Table 4).
Additionally, a positive association between mean fish length and salinity, TDS, conductivity, ammonia, and pH was found (r = 0.901, p = 0.000, r = 0.909, p = 0.000, and r = 0.850, p = 0.001, respectively). While there was a negative association between mean shrimp weight and DO (r = –0.637, p = 0.035), alkalinity (r = –0.894, p = 0.000), and temperature (r = –0.393, p = 0.22).
The eight physicochemical characteristics explained 97.4% of the difference in shrimp weight for T2, while for the other two treatments, the percentages were 94.9% and 95.1% for T1 and control, respectively. The results of the multiple regression analysis produced distinct R2 values for each of the three ponds, as shown in Table 5.
From pond preparation till harvesting, 140,000 Tk was spent. The highest output was 832.8 kg/ha in T1 (20% N. indica). Table 2 shows greater survival at T1. Big size meant higher price than other treatments. Big shrimp cost 650 Tk/kg. Based on this price, profit was 401,320 Tk/ha. Cost-benefit analysis of shrimp culture with N. indica in a 1 ha pond is shown in Table 6.
| Particulars | Treatments | ||
|---|---|---|---|
| Control (without Najas) | T1 (20% of Najas) | T2 (40% of Najas) | |
| Total variable costs (BDT) | 130,000 | 130,000 | 130,000 |
| Total fixed costs (BDT) | 10,000 | 10,000 | 10,000 |
| Production | |||
|---|---|---|---|
| Yield (kg) | 397.86 | 832.8 | 641.62 |
| Price (BDT/kg)1) | 500 | 650 | 600 |
| Economic analysis | |||
|---|---|---|---|
| Total production costs (BDT) | 140,000 | 140,000 | 140,000 |
| Gross return | 198,930 | 541,320 | 384,972 |
| Net profit | 58,930 | 401,320 | 244,972 |
| Benefit/ cost ratio | 0.42 | 2.86 | 1.74 |
Discussion
In order to boost shrimp farm productivity, water quality management is undoubtedly a crucial factor. The DO level was highest in T1 (8.55 ± 1.06 mg/L) and lowest in control (6.75 ± 1.33 mg/L), with a statistically significant difference (F = 7.23, p = 0.025) between the two. Gupta et al. (2004) stated in their study that maintaining DO within the range of 3–10 mg/L is important. The DO level in our experiment was beside the higher limit, it may be occurred due to two reasons. In our experimental ponds exchange of water with tidal water from nearest Daratana river, fortnightly and in treatment ponds photosynthesis occurred due to availability of N. indica. For penaeid shrimps, optimum value of DO for maximum growth rate is 6 ppm. The recorded temperature (30.88 ± 1.12°C – 31.04 ± 1.1°C) was much identical with the recommended ideal range by Krishnani et al. (2006), for the majority of brackish water penaeid shrimp, 28°C to 32°C is the ideal temperature. The prevailing consensus is that pH of water 7.5 to 9.0 is more suitable for shrimp growth. The pH level in the three ponds varied slightly and significantly between N. indica containing pond and the control pond (p = 0.160), but they were within the acceptable range as suggested pH ranging from 7.5 to 9.0 is typically regarded as suitable for shrimp production were within the allowed range of 6.0–9.0 for estuarine and marine ecosystems, indicating that they were close to the pH range (Boyd, 1982).
The salinity of water ranged from 4.49 ppt to 5.9 ppt in all of the ponds and it was lowest during culture period. However, these findings are slightly relevant to Chandrakant (2003), brackish water shrimp farms should have salinity levels between 10 and 40 ppm. Salinity was lower in our culture ponds.
In comparison to control ponds, the mean values of total alkalinity with the ranges of (71.0 ± 5.04–86.11 ± 6.93 mg/L) were significantly different in ponds containing N. indica. But were within the range that was proposed by Boyd (1982), and they correspond to other authors (Rahman et al., 2015). The measured ammonia level (0.02 ± 0.06–0.04 ± 0.08 mg/L) was within the desirable range according to research’s outputs of Krishnani et al. (2006).
Compared to control ponds, ponds with N. indica demonstrated better growth performance in terms of length and weight gain. Up to the first two weeks of the grow-out stage, all three treatments shown comparable development. Thereafter, however, their growth differed until the study’s conclusion. More specifically, the T1 ponds shrimp consistently showed a rank-first growth trend over T2 and control (Fig. 1). T1 (30.44 ± 0.77) showed greater growth than T2 (26.21 ± 0.85) and the control (17.38 ± 0.63), respectively. It’s possible to say that the presence of N. indica had a significant (p < 0.05) impact on how growth metrics fluctuated. In comparison to other treatments, the T1 demonstrated a considerably (F = 66.780, p = 0.000) greater final mean BW.
Shrimp from T1 had a BW that was nearly 42.9% and 13.2% higher than shrimp from T2 and the control, respectively. The results align with the findings of Ma et al. (2020), which also shown that the growth performance of macrobrachium rosenbergii improved significantly when aquatic plants retain 15% in total waterbody. Additionally, Ali et al. (1999) found that shrimp growth is impeded and its maximum potential is not realized when aquatic plants are maintained at a 40% concentration in the total waterbody.
T1 demonstrated a greater correlation in the length-weight connection than T2 and the control, despite the fact that their ‘r’ values were not substantially different. The SGR value at harvesting (8.4%, 9.02%, and 8.86%/day) varied substantially among the three treatments (F = 4,000.66, p = 0.000), but was largely consistent with the findings of Susilowati et al. (2014).
Therefore, it is reasonable to assume that N. indica may produce various FCRs under the appropriate environmental conditions. T1 outperformed other treatments in terms of gross yields and survival, which is similar to some of the work done by Ma et al. (2020). It can be because the pond contains 20% N. indica. However, T2 demonstrated lesser output than T1, which may be compared to the findings of Izzati (2011) and Ali et al. (1999). The remaining parameters in T1, T2, and control had the more or less same findings. According to Izzati (2011), co-cultivating tiger shrimp with the seaweed Gracilaria was superior for promoting shrimp growth since it was the most effective bio-filter and could reduce nutrient excesses, improving water quality and providing shrimp with food. The growth exponent ‘b’ value ranged from 3.26 to 3.37 for all ponds, demonstrating a close-to-allometric development trend and uniform shrimp morphology. However, according to Spiegel (1991), the control (3.37), T2 (3.33), and T1 (3.26) displayed favorable allometric growth patterns (Table 3).
The SGR dropped correspondingly as physico-chemical parameters fluctuated a little bit over the course of the culture period. However, the gained SGR of T1, which corresponded to a DO level of 8.55 ± 1.06 mg/L, was really higher (Fig. 3). Therefore, it is evident that elevated DO levels had a favorable impact on shrimp growth. Additionally, Ekubo & Abowei (2011) cautioned that fish are likely to perish if DO falls below 0.3 ppm for an extended period of time. A concurrent rise in temperature and an increase in SGR were observed. The greatest SGR of 16.85%/day for T1 was observed at a temperature of 30.8°C (Fig. 3), which matched the range by which it was measured (Rahman et al. 2015).
The greatest SGR in the current investigation was likewise obtained in T1 (Fig. 3) at pH 8.1 (near neutral), salinity 2.98 ppt, and ammonia 0.02 mg/L, all of which were below the 0.2 mg/L ammonia level that is advised for shrimp culture in ponds. Boyd (1982) identified an appropriate pH range for shrimp cultivation ranges between 6.5 and 9, which is consistent with the pertinent study findings. The maximum survival rate was observed at T1 (68.4%). This is due to the fact that the plants provide them with a sufficient amount of shelter and allow them to move freely. Consequently, when molting occurs, they can easily conceal themselves within the plants in order to protect themselves from cannibalism by others.
The regression coefficient’s result (R2 = 0.974–0.949) shows that factors affecting water quality had a significant impact on shrimp growth. Tho et al. (2011) revealed a substantial positive association between mean weight and length and water quality indicators, which is consistent with our results. The slope of a regression line is determined by the partial regression coefficient, together with its magnitude (b-value). In this study, the b-values for temperature (–0.185) and alkalinity (–0.348) had negative signs, indicating that the regression equation predicted a drop in shrimp weight (the dependent variable) of 0.185 g and 0.348 g, respectively, for every rise in one unit of DO and NH3. The b-values for DO, pH, salinity, TDS, conductivity, and ammonia all had positive signals, indicating that there was a proportional rise in shrimp weight for every unit that these parameters increased. For instance, the regression equation projected that for every unit of DO increase, shrimp weight would rise by 0.107 g. Shrimp culture with the presence of N. indica will be the profitable. In this farming highest BCR was found 2.86 that clearly indicate it can be a profitable culture system for the farmers.
Growth and production-wise, T1 was superior to both the control group and T2. Technically feasible, the introduction of N. indica with tiger shrimp (P. monodon) cultivation improves water quality and increases yield. The shrimp often feed on soft plant tissues that are home to tiny organisms including copepods, ostracods, insect larvae, nematodes, and snails. Their primary emphasis is on the decomposing vegetation that covers the floor of the pond. As a result of their burrowing propensity, shrimp prefer clean, soft bottoms; nevertheless, an overabundance of aquatic vegetation at the substrate could be detrimental to the development and reproduction of shrimp. In conclusion, it can be asserted that the controlled cultivation of shrimp with N. indica enhanced shrimp production and economic advantages for producers.







